Pfizer stopped selling Depo-Testosterone in Canada in 2025, ending what had been one of the country’s main sources of injectable testosterone since the 1950s. The company hasn’t provided any details beyond “business reasons,” but the timeline leading up to the exit helps explain why it happened.
With Pfizer gone, Canada now relies on just two manufacturers for injectable testosterone. That leaves an already fragile supply system vulnerable, with limited ability to absorb disruptions if one product goes into shortage.
How Depo-Testosterone Disappeared from the Canadian Market
Depo-Testosterone went into shortage in July 2021 and stayed there for nearly two years. During that time, Pfizer confirmed it had redirected manufacturing capacity to produce its COVID-19 vaccine, which likely caused the disruption.
When Depo-Testosterone returned in 2023, supply didn’t fully stabilize. Availability remained inconsistent, and pharmacies didn’t always have it on hand.
This wasn’t just a problem with one product. The injectable market had already been unstable for years. Bausch’s testosterone enanthate product, Delatestryl, which was on the market until 2024, went into shortage repeatedly, sometimes multiple times in a single year. Patients often had to call different pharmacies, transfer prescriptions, and track down their usual product. When that failed, they were forced to switch to a different testosterone product.
By late 2024, the situation tightened further. Taro, the only generic version of testosterone cypionate in Canada, went into shortage for over a year. That left fewer options and put more pressure on the remaining supply, including Pfizer’s Depo-Testosterone.
Around this time, Pfizer appears to have made the decision to exit. Depo-Testosterone was marked “cancelled post market” in July 2025, before the discontinuation was formally reported in October.
These events show a market that had been unstable for years, with repeated shortages across multiple products. Pfizer’s exit came after a period when supply had already become difficult to maintain, further reducing the system’s ability to handle disruptions.
Why Pfizer Left Canada
Depo-Testosterone is still sold in the United States. The product hasn’t been discontinued globally.
That points to a more specific conclusion: Pfizer chose to leave the Canadian market.
Injectable testosterone is not a simple product to maintain. It requires sterile manufacturing, dedicated production capacity, and ongoing supply management. At the same time, it is an off-patent drug with limited pricing flexibility, which puts a ceiling on how much revenue it can generate.
Those constraints make the underlying economics important. The United States has a much larger patient base and more flexible pricing, which makes it easier to sustain a product like this. Canada is smaller and more tightly priced, which limits the total return a company can expect, even when demand is steady.
In that context, a product like Depo-Testosterone can remain viable in one market and not in another.
A Pattern of Companies Abandoning Injectable Testosterone in Canada
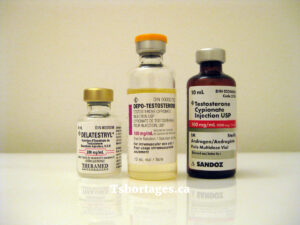
There’s no detailed public explanation for Pfizer’s decision beyond “business reasons,” but it isn’t an isolated case. In the past 15 years, multiple companies have stopped selling injectable testosterone products in Canada, including Theramed, Sandoz, Valeant, and Bausch.
Injectable testosterone requires consistent manufacturing and supply, but offers limited flexibility in pricing. That combination can be difficult to sustain, especially in Canada’s smaller market where total volume is limited.
Repeated shortages in recent years have made supply harder to manage. When one product becomes unavailable, patients and pharmacies shift to whatever remains, quickly putting that supply under pressure as well. That kind of demand volatility makes it harder for manufacturers to maintain stable supply and plan production over time.
Pfizer’s exit follows that same pattern.
What Pfizer’s Exit Means for Testosterone Supply in Canada
For a brief period after 2023, Canada had three injectable testosterone products on the market: Pfizer and Taro supplying cypionate, and Bausch supplying enanthate. That level of overlap hadn’t happened since 2012. It gave the system some flexibility, allowing demand to shift between manufacturers when shortages occurred.
That’s no longer the case.
Canada now has two injectable options, cypionate and enanthate, supplied by two manufacturers: Taro and Hikma. This reduces overall system flexibility, and overall supply remains limited.
As a result, shortages play out differently. When supply is disrupted, there is limited ability to absorb demand across products. Problems with production, distribution, or inventory can still affect overall availability, especially if one product goes into shortage and demand shifts to the other.
In practice, that means fewer alternatives when shortages happen. Switching between the two products might not be possible, and pharmacies have less ability to source around gaps. Shortages can still last longer if demand shifts quickly from one product to the other and puts pressure on limited supply.
Pfizer’s exit exposed how vulnerable Canada’s testosterone supply is. With only two manufacturers of injectable testosterone, an already strained supply is prone to disruption, and Canada has limited backup when one product goes into shortage.